Rinodina cana
|
|
|
|
Family: Physciaceae
[Rinodina arenaria var. cana Arnold, moreRinodina iowensis Zahlbr.] |
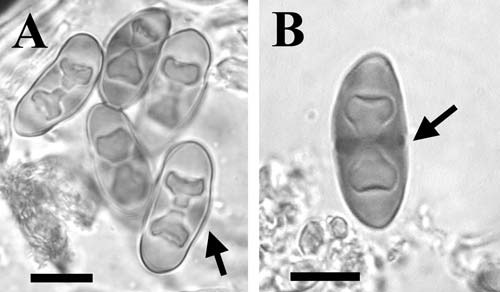
MB#404248 Basionym. Rinodina arenaria var. cana Arnold, Verh. K.K. Zool.‑Bot. Ges. Wien 22: 289 (1872). Rinodina iowensis A. Zahlbr. Ann. Naturhist. Hofmus. Wien 20: 40 (1905). Exsiccatae. Arnold Lich Exs. 494 (K in BM). Description. Thallus thin, dark grey to grey-brown, rimose-areolate; areoles often sharply angular, to 0.60-1.00 mm wide, margins sometimes raised; surface plane and matt; margin indeterminate or determinate; prothallus absent or dark, fimbriate; vegetative propagules absent. Apothecia innate, rarely becoming broadly attached, frequent, mostly one per areole, therefore rarely contiguous, to 0.30‑0.50 mm in diam.; disc black, persistently plane; thalline margin usually absent. Apothecial Anatomy. Thalline exciple rarely distinguishable from thallus, then ca. 40 µm wide; cortex 10-20 µm deep; epinecral layer absent; crystals absent in cortex and medulla; cortical cells to 4.5-6.0(-7.5) µm wide, usually pigmented; algal cells to 11.5-14.5 µm long; proper exciple hyaline, (5-)10-15 µm wide laterally, expanding to 15-25µ above, sometimes with Bagliettoana-green; hypothecium colourless, 40-90 µm deep; hymenium 70-100 µm high, not inspersed; paraphyses 2.5-3.0 µm wide, usually conglutinate, apices to 4.5-6.0 µm wide, lightly pigmented, immersed in dispersed pigment forming a light, orange-brown epihymenium; asci 55-65(-75) x 20-25 µm. Ascospores 8/ascus, Type A development, Mischoblastia-type, (16.0-)19.0-20.0(-23.0) x (8.5-)10.5-11.0(-13.0) µm, average l/b ratio 1.7-1.9, lumina rather angular, sometimes becoming rounded (Pachysporaria-like); torus poorly developed or narrow, dark at maturity; walls not ornamented. Pycnidia not seen; conidia bacilliform, 3.5-4.0 x ca. 1.0 µm (in type description of R. iowensis). Chemistry. Spot tests all negative; secondary metabolites not detected (Heklau et al. 1981; Giralt 2001). Substrate and Ecology. Mainly recorded on granite boulders, also on sandstone, in xeric habitats and shaded chert. The only recorded elevation, and probably the highest, is at 1,555 m in Big Bend Nat. Park, Texas. The species has been recorded three times accompanied by R. oxydata. Distribution. The distribution of R. cana as presently known is centred in Illinois but is also known from Louisiana and Texas. Northern outliers occur in Minnesota and Connecticut. A record for Wisconsin (Thomson 2003) has not been confirmed. The first correct report for North America is that of Wilhelm (1998) from DuPage Co., Illinois. In Europe it is known from the southern Alps to southern Spain (Mayrhofer 1984, Giralt 2001). Mayrhofer et al. (1990) reported R. cana for the first time from North America. On reexamination, this specimen in MICH (Fink 1072, Rainy Lake City) has been found to possess atranorin crystals, in variable quantities, in the cortex and spores > 20 µm long. It therefore belongs to R. destituta. Fink (1935) lists R. iowensis as a synonym of R. ocellata (Hoffm.) Arnold (syn. R. lecanorina (A. Massal.) A. Massal.) which does not occur in North America. Notes. Rinodina cana is distinguished from R. oxydata by its lack of atranorin, darker thallus, innate or cryptolecanorine apothecia, and Mischoblastia-type spores with an atypical structure. The lumina are more angular and possess broader canals than is usual for the spore type and were compared to the Pachysporaria-type by Mayrhofer and Poelt (1979) because the lumina tend to become rounded with age. Giralt (2001) on the other hand assigns the spores to Milvina-type on the basis of the rather angular lumina throughout much of the development sequence but the walls are very lightly pigmented for this spore type. There is no doubt that the spores of this uncommon species are difficult to classify but in the author’s opinion they are closest to the Mischoblastia-type. The variable presence of Bagliettoana green in the proper exciple would tend to support this opinion since the pigment is most commonly associated with this spore type. The often sharply angular areoles, innate apothecia and dark thallus colour are characters reminiscent of the Buellia aethalea agg. In fact, the lectotype of R. iowensis is accompanied by a specimen belonging to this group of species, possibly Buellia nigra (Fink) Sheard (1969). North American material is indistinguishable from European specimens studied with the exception that pruinose discs have not been observed. Giralt (2001) notes that, in section, the epihymenium crystals dissolve in KOH. Such crystals have been noted in some other species with conglutinate paraphyses and seem to be due to the extrusion of hymenial gelatin. Specimens examined. U.S.A. CONNECTICUT. Litchfield Co., Goshen, 1886, H.A. Green (PH). ILLINOIS. DuPage Co., Morton Arboretum, G. Wilhelm 19638; LaSalle Co., Covel Creek, M. Jones 1764; Marshall Co., Hopewell Estates, M.D. Jones 3996 (all MOR); Randolph Co., Castle Rock, A.C. Skorepa 2552 (WIS); Piney Creek Ravine, A.C. Skorepa 6221 (BALT); Union Co., Pine Hills, A.C. Skorepa 483, 484 (WIS); Will Co., Lockport Prairie, R.D. Hyerczyk 1767; Rock Run Greenway, R.D. Hyerczyk 1752 (both personal herb.). IOWA. Fayette Co., Fayette, B. Fink (BM, FH, US); 1895, B. Fink (CANL, DUKE, FH, MIN, MSC, MU, NY, UC, UPS, US, WIS); 1897, B. Fink (US). KANSAS. Anderson Co., 2mi E Welda, C.A. Morse 15146; McPherson Co., Canton, 6.5mi NW, C.A. Morse 13256; Saline Co., Brookville, C.C. Freeman 21693 (all KANU). LOUISIANA. Natchitoches Co., Red Dirt Wildlife Management Area, S.C. Tucker 24646 (SBBG); Vernon Par., 3.5 mi NW Hornbeck, C.M. Wetmore 17521, 17541 (ASU, MSC, WIS). MINNESOTA. St. Louis Co., Headlight Island, C.M. Wetmore 32414 (MIN). TEXAS. Brewster Co., Big Bend Nat. Park, C.M. Wetmore 18440 (MSC); Robertson Co., SE Hearne, S.C. Tucker 15861 (LSU). Selected References. Mayrhofer & Poelt (1979), Mayrhofer (1984a), Giralt (2001 Plate X: A), Thomson (2003). |